Alien flora and fauna are a staple part of much science fiction. A large body of literature exists describing ways of building plausible worlds. When it comes to 'critters' one is left a little more to one's own devices.
The purpose of this series of articles is to consider relevant aspects of biology, (bio)chemistry, physics, physiology and ecology in order to flesh out game statistics and build better aliens!
[The rules presented in the various incarnations of Traveller and some other RPGs (I haven't sighted a copy of GURPS Bestiary or Uplift) present a good starting point for creating creature encounters (approximate ecological niche, size, speed, armour, health, damage capacity, etc.)]
A Definition of 'Life'
For the purposes of this discussion, 'living things' are those systems which
- are capable (to varying extents) of growth, self-repair and reproduction
- require various energetic and chemical inputs to enable these processes.
These criteria get blurry at both ends of the size scale: is a prion 'alive'? a single enzyme? an ecosystem?
Leaving this philosophical problem aside, let us begin.
Prequisites
Three basic conditions seem to be necessary to the development of life.
- Building blocks
- Energy
- Solvents
1. Building Blocks
Appropriate elements need to be present for anything to happen at all.
Cosmic abundance of the first thirty elements:
| Hydrogen |
1 |
Chromium |
9x10-7 |
| Helium |
0.25 |
Manganese |
5x10-7 |
| Oxygen |
10-4 |
Phosphorus |
2x10-7 |
| Carbon |
~5x10-5 |
Chlorine |
" |
| Neon |
" |
Potassium |
10-7 |
| Magnesium |
" |
Titanium |
9x10-8 |
| Silicon |
" |
Cobalt |
7x10-8 |
| Iron |
" |
Fluorine |
5x10-8 |
| Sulphur |
2x10-5 |
Zinc |
" |
| Nitrogen |
7.5x10-6 |
Copper |
2x10-8 |
| Nickel |
" |
Vanadium |
10-8 |
| Aluminium |
5x10-6 |
Boron |
5x10-9 |
| Sodium |
2x10-6 |
Scandium |
10-9 |
| Calcium |
" |
Beryllium |
10-10 |
| Argon |
10-6 |
Lithium |
10-11 |
Heavier elements are RARE.
In the 1960s, Urey and Miller demonstrated that the four basic classes of terrestrial 'biological molecules', (amino acids, fats, sugars and nucleotides) could be generated from a mixture of water, methane (CH4), ammonia (NH3) and hydrogen subjected to electricity.
Carbon provides a skeleton on which complex compounds can be built; it is tetravalent and forms strong stable covalent bonds (electron sharing) with other elements.
Oxygen is a highly reactive chemical which permits the development of polar species. Its higher affinity for electrons leads to localised negative charge around the oxygen atom - the donor becomes relatively positively charged. Water molecules are a good example:
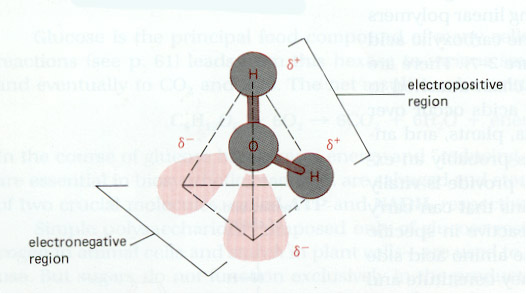
Nitrogen can form a wide variety of compounds with the other elements; ammonia is an important solvent.
Are there any other candidate elements for the 'short list of building blocks'?
Silicon: like carbon, it is tetravalent and therefore has the potential to form complex structures. However, silicon has a larger covalent radius (bigger atoms!) and therefore compounds with pure silicon backbones (e.g. the silanes (SiH4, Si2H6, etc.)) are not as stable as their carbon analogues.
In fact silanes larger than SiH4 decompose explosively in water and oxygen; they may be more stable in less polar solvent systems or at extremely low temperatures, neither of which are conducive to the development of the complex chemical systems we are looking for.
Silicones (chains of alternating silicon and oxygen atoms) are very stable and represent a viable (but unusual) alternative to carbon as a 'foundation material'. [Silicates - silicon oxides - comprise much of the mass of rocky bodies!]
Boron: It has a similar covalent radius to carbon, so compounds are likely to be more stable. Being trivalent, it can replace nitrogen in most carbon compounds. Like silicon, boron hydrides (B2H6, etc.) react vigorously with water and oxygen. Compounds of boron and nitrogen would either be explosively unstable or crystalline solids.
Other elements reflect the metals and salts present in the primordial solvent. The following elements are also present in varying proportions in terrestrial life (in no particular order):
Chlorine, phosphorus, sulphur, fluorine, selenium, iodine, sodium, potassium, magnesium, calcium, iron, zinc, copper, cobalt, manganese, molybdenum, and vanadium.
Many of the metallic elements listed above have catalytic functions. The non-metals could be important in the formation of solvents and metabolic pathways.
An important caveat: The amount of energy available will determine local temperature and atmospheric composition. This limits the variety of possible building blocks for surface life.
As temperature increases, the kinetic energy of all molecules increases. Gas molecules may get fast enough to escape the gravitational pull of a world. Molecules with greater average speed than the world's escape velocity will only be present in trace amounts. Those with average speeds over about 10% of escape velocity will be lost over the course of a few million years.
Escape Velocity (m/sec) = sqrt(2 * G * R)
where G is surface gravity (1G=10ms-2), R radius in metres. Surface gravity can be determined as per the world generation rules you are using. It is a function of planetary mass and size.
For the earth, escape velocity = sqrt(2x1x6.4x106) = 3578 m/sec
Gas molecule velocity (rms velocity in m/sec):
v (m/sec) = sqrt(24936T/n)
where T is absolute temperature and n molecular mass of gas concerned (H2 = 2, O2 = 32, etc.)
Multiply the rms velocity by 0.817 for most probable velocity; by 0.922 for mean velocity
At 288K (25C) : rms velocity of an oxygen molecule = 474 m/sec; rms velocity of a hydrogen molecule = 1895 m/sec
So most oxygen is located in the lower layers of the Earth's atmosphere and hydrogen is only present at high altitude (a small amount is present in the uppermost layers of the atmosphere - 600+km altitude).
(Most of the hydrogen present on Earth is 'locked up' in water or organic compounds; and the atmospheric component has long since attained escape velocity by absorbing solar radiation).
2. Energy
The Second Law of Thermodynamics implies that a chemical system will tend to move towards the lowest energy state. Living things rely on energy from "outside" to maintain internal complexity and order.
The most important source (at least for surface-based life) is from the star(s) the planet orbits.
The 'solar constant' is ~1353W per square metre at one AU (150 million km); it varies inversely with the square of the distance from the primary, directly with luminosity, and is attenuated by the atmosphere, depending on its composition (roughly inversely with atmospheric pressure/density; the maximum energy (100% efficiency) obtainable on the ground on Earth is 956W per square metre).
Internal heat, from the decay of radioisotopes in the mantle and core regions of metallic and rocky worlds (in GURPS Traveller: First In terms, non-rockballs) leads to familiar phenomena such as plate tectonics and vulcanism. As recently demonstrated, the deep ocean vents allow the development of a vigorous ecosystem in the vicinity of the plume.
Satellites of gas giants develop a significant amount of internal heat from tidal effects driven by their primary. Io is a good example of this. Europa may have large bodies of liquid water beneath its icy exterior.
Other compounds may be present that make important reactions more likely to happen (catalysts). The required activation energy may be reduced by the presence of intermediary electron donors (e.g. metals and metal ions) or a substrate which allows compounds to interact more readily (e.g. clays, porous rocks, etc.)
3. Solvents
Chemical reactions need an appropriate medium to take place in. Solvents need to have certain properties for our purposes.
- They should be composed of common elements from the list above.
- They should be liquid across a wide temperature range - this increases the probability of 'life processes' starting in them in the first place.
- They need to be relatively polar to encourage the dissociation of salts into their constituent ions; this facilitates the transfer of electrons and thus enables the development of potential differences. This becomes important in a wide variety of cellular functions (to be discussed in a later post).
Some candidate solvents:
|
Solvent |
Formula |
Melting Point |
Boiling Point |
Dipole moment |
| Water |
H2O |
0 |
100 | 6.2 |
| Hydrogen peroxide |
H2O2 |
-0.4 |
158 | 7.3 |
| Ammonia |
NH3 |
-78 |
-33 | 4.9 |
| Nitric acid |
HNO3 |
-47 |
86 | 7.2 |
| Nitrosyl chloride |
NOCl |
-65 |
-6 | 6.3 |
| Hydrazine |
N2H4 |
1 | 114 | 5.8 |
| Hydrogen fluoride |
HF |
-83 | 19 | 6.1 |
| Sulphur dioxide |
SO2 |
-73 |
-10 | 5.5 |
| Sulphuryl chloride |
SO2Cl2 |
-54 |
69 | 6.0 |
| Thionyl chloride |
SOCl2 |
-104 |
75 | 4.8 |
| Sulphuryl fluoride |
SO2F2 |
-120 |
-52 | 3.7 |
| Phosphorus trifluoride |
PF3 |
-160 |
-95 | 3.4 |
| Phosgene |
COCl2 |
-104 |
8 | 3.9 |
| Hydrogen chloride |
HCl |
-114 |
-85 | 3.6 |
| Hydrogen sulphide |
H2S |
-83 |
-62 | 3.2 |
Melting and boiling points are in degrees Celsius; boiling points assume one atmosphere (101 kPa, 14.5psi or 760mm mercury/Torr) ambient pressure. Dipole moment is a measure of molecular polarity and is expressed in units of 10-30 coulomb metres. Salts are obviously much more polar; for example sodium chloride has a dipole moment of 30.
The two most likely compounds are water and ammonia, on the basis of cosmic abundance and their intermediate reactivity, but the others are certainly plausible.
Likely Worlds (using G:T 'First In')
Given the criteria above:
- Surface based life requires an atmosphere to prevent important chemicals from boiling off into space.
- Underground life requires active geology or tidal heating.
There is an enormous degree of latitude possible with (underground) life, especially considering more exotic solvents of freezing points of -100 degrees Celsius or lower! This gives us plenty of world types to play with. In fact almost any world could support some sort of subsurface microbial life, given the right chemistry.
Worlds with surface liquid are much more likely to be life bearing. This liquid will be the solvent system in which life arises, on balance of probabilities.
- Worlds with oxygen or chlorine containing atmospheres must support some form of life. These two elements are so reactive that they would ordinarily be locked up in rocks or salts. The only other way that large amounts of gaseous oxygen or chlorine could be present would be violent geological activity.
What? No cryogenic creatures or plasma beings?
I have ignored the possibility of creatures that function at extremes of temperature due to the fact that reaction rates will either be very slow or terribly fast depending on which end of the scale is being considered.
The other problems with high temperatures are that very few compounds remain stable; certainly complex molecules are unlikely to survive. Their formation in any case is problematic as there is plenty of energy available but not enough time for reactants to stick together.
Low-temperature creatures (superconducting nervous systems?) need highly concentrated building blocks in order to happen at all (again, consider the composition of typical superconductors). In summary: high/low temperature = high/low energy = low probability of establishing the necessary level of order.
Aside : Eutectic mixtures and temperature ranges:
A eutectic mixture is one whose melting point is lower than any of its ingredients.
This effect is a consequence of freezing point depression - one of the colligative properties of a solution. (Those properties depending on the amount of substance dissolved in a solvent rather than the identity of the solute - the others are boiling point elevation, vapour pressure depression and osmotic pressure).
An example: water's freezing point can be depressed by 1.86 degrees C per mole of substance added per kilogram of water. Ammonia has a solubility of 480g ammonia per kilogram of water at 25 degrees C. The mass of one mole of ammonia is 17g (NH3) - so 480g is 28.2 moles. The freezing point of our saturated ammonia-water solution is roughly -50 degrees Celsius!
Next part: From Primordial Soup to Cells
 Freelance
Traveller
Freelance
Traveller